Genetic testing can greatly improve the chance of achieving a successful pregnancy. Our fertility experts outline the advantages and disadvantages of preimplantation genetic testing below.
What is the difference between the different types of genetic testing?
Genetic testing of embryos is used for a number of different things, and we’ll talk a little bit about the different forms of preimplantation genetic testing, or PGT, in just a moment. But in general, genetic testing of embryos is done at the early stage of development when embryos are blastocysts.
At that stage, there are two distinct cell populations. The trophectoderm, which forms the placenta, and the inner cell mass, which forms the baby. What is done is about 5-8 cells out of several hundred cells are removed from the trophectoderm, which will eventually form the placenta without harming the embryo’s ability to create a child, and those cells are utilized for genetic testing for various purposes.
In general, PGT is broken into three separate categories. There is PGT-A (preimplantation genetic testing for aneuploidy), PGT-M (preimplantation genetic testing for monogenetic diseases), and PGT-SR (preimplantation genetic testing for structural rearrangements).
Preimplantation genetic testing for aneuploidy (PGT-A)
PGT-A is utilized to test whether or not the embryo has the right number of whole chromosomes.
All of us have 46 chromosomes, and those chromosomes are created when the egg goes from a whole set of DNA to a half set, and the sperm does the same. They combine to create a new child.
That process, sometimes, doesn’t go correctly. There can be a tendency to throw out too many chromosomes or keep too many chromosomes. When that happens, three main outcomes can occur.
- The embryo never implants and creates a pregnancy, and that’s actually the most common outcome.
- The pregnancy implants and creates a pregnancy that ultimately, unfortunately, results in an early first-trimester miscarriage.
- There are a couple of chromosome abnormalities that can result in ongoing pregnancies. For example, Trisomy 21, also known as Down syndrome. So, PGT-A tests for whether or not an embryo has the correct number of whole chromosomes within it, and that allows for a better chance that that embryo, when transferred to the uterus, will create a healthy child.
Preimplantation genetic testing for monogenic diseases (PGT-M)
Preimplantation genetic testing for monogenic diseases, or PGT-M, is a special type of genetic testing that includes PGT-A but also looks for a specific genetic abnormality.
These might be found on your expanded carrier screen, which is done as part of the workup. And that looks for, genetic diseases inherited from the egg and the sperm. This would be like, for example, cystic fibrosis.
A genetic mutation in the DNA causes cystic fibrosis, and if both the egg and the sperm source carry a mutation for cystic fibrosis, there is a 1 in 4 chance of having a child affected by the disease.
If we know what we are looking for, PGT-M is able to screen embryos and determine whether or not embryos are unaffected by the condition, whether or not they are carriers of the condition, just like the egg or the sperm source, or whether the embryo is affected by the genetic disease.
That allows us to select an embryo that is either a carrier or unaffected and transfer that embryo into the uterus in the hopes of creating a child that is not only genetically normal with PGT-A but is not affected by the single gene condition when utilizing PGT-M.
Preimplantation genetic testing for structural rearrangement (PGT-SR)
PGT-SR has to do with when the egg or the sperm source has the right amount of DNA, but that DNA is in an abnormal orientation.
Now what happens, is when the egg or the sperm source goes from a whole set of DNA to a half set of DNA to combine with the other, resulting in a much higher likelihood of a genetic imbalance in the embryo.
That, with PGT-SR, ensures that the embryo has the right amount of DNA so that when transferred, we can have it be either a genetically normal embryo or a genetically balanced embryo – just like the egg or sperm source would have been.
Gender Selection – Choose the sex of your child with genetic testing
As part of PGT-A, we test for all of the chromosomes. As part of that information, we obtain information about the sex chromosomes. The X and the Y. So, XX is female and XY is male.
There are several different medical reasons which might cause an individual or clinician to select one gender or the other. One of the potential causes is an X-linked genetic mutation, and in that case, we may select female embryos over male embryos.
Some of them might be a Y-linked genetic mutation and in which case we may end up choosing one embryo over the other.
One of the things that we see with PGT-A is that we get information about the genders of the embryos. This information is part of the patient’s record, and it is given to the patient to do with what they choose. By doing so, they may end up deciding to transfer one gender over another.
Genetic Testing Research
When it comes to genetic testing of embryos, it’s an area of the infertility field that is rapidly evolving. In fact, RMA has been a part of the only non-selection study with the use of next-generation sequencing, or NGS, which is the most common platform utilized for genetic testing presently.
Now, I want to explain briefly what a non-selection study is and why that is so important.
A non-selection study is really the only way that you are able to determine the positive and negative predictive values of the tests that you utilize. One of the things that many people are concerned about is, “Gosh, what if I’m throwing away an embryo that ultimately could create a child?”
Well, this is how a non-selection study answers that question. In our non-selection study, patients underwent IVF treatment and had a biopsy of the embryo, but that biopsy result was not tested. The biopsied cells were held until an embryo transfer was done, and the outcome of that embryo transfer was known.
There are only three outcomes:
- a healthy live birth
- a miscarriage
- no pregnancy occurred at all
We then ran the genetic testing results to see if the genetic testing results accurately predicted the outcome that was actually seen.
What we ended up finding was that several hundred embryos were transferred that were actually deemed to be genetically abnormal with the test. Not a single one of them resulted in an ongoing pregnancy. So, that tells us that this test has very, very good accuracy.
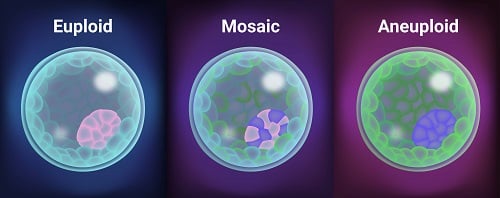
One of the other interesting things found in this study was that of mosaic embryos. Mosaicism is one of the most challenging areas in the field of genetic testing of embryos at the present time. With the advent of newer genetic testing platforms, this new diagnosis of mosaicism (when some cells are normal and some cells are abnormal in the same biopsy specimen) was able to be diagnosed. At that time, we didn’t really know what to do with this diagnosis.
As part of this non-selection study, embryos that were ultimately deemed mosaic were also transferred. And information that was also presented at the American Society for Reproductive Medicine in 2022 looked at this and found that mosaic embryos actually had fairly equivalent pregnancy outcomes to whole chromosome normal embryos.
All this is to be said that RMA is on the cutting edge of genetic testing of embryos. We are constantly striving to answer new questions that arise, and we do so with the proper studies, with the use of non-selection studies, and that is really the only way that you can truly know what the outcome is for the embryos that are being tested and to know that you are getting accurate results.
Who should get genetic testing?
All patients who are looking to conceive should be offered genetic testing.
A consultation with a genetic counselor can identify whether the couple is at an increased risk of having a baby with a genetic disease.
Individuals with strong family histories of inherited diseases or specific cancers should also seek a consultation with a genetic counselor to see if there is a genetic cause.
When is it safe for genetic testing (PGT) to be performed on an embryo?
Following fertilization, embryos progress through multiple stages of differentiation.
A day 3, or cleavage-stage embryo, consists of eight cells called blastomeres, each of which has the potential to develop into a placenta and or fetus.
Day 5 embryos, or blastocysts, are made of approximately 200 cells and have clearly differentiated which cells are destined to be the placenta (trophectoderm) and which will become the fetus (inner cell mass).
PGD involves the removal of cells from a single embryo to assess the genetic makeup of that embryo.
Biopsy of a day three embryo involves the removal of 1 or 2 blastomeres from an eight-cell embryo, whereas day five trophectoderm biopsy involves the removal of a few cells from the placental portion of a 200-cell embryo.
Therefore, day three biopsies require the removal of a greater proportion of the total embryo mass.
Moreover, because each cell is less differentiated on day three, the cells removed from a day three embryo may have otherwise contributed to the formation of the fetus.
By contrast, day five trophectoderm biopsy removes a much smaller proportion of the overall embryo mass, and those cells are exclusively removed from the placental compartment.
Research performed at RMA and published in a peer-reviewed journal (Fertility & Sterility) demonstrates that day three biopsy is harmful to embryos, resulting in a 39% reduction in the embryo’s chance of becoming a healthy baby.
Obtaining a small amount of genetic DNA from the trophectoderm at the blastocyst stage (day 5) did not impair the embryo’s ability to implant.
Furthermore, trophectoderm biopsy has been shown to be more accurate than cleavage-stage biopsy because there is more DNA for genetic analysis.
In conclusion, doing genetic testing on embryos on day 5 is the safest and more accurate time to perform genetic testing of an embryo.
What conditions and or diseases can be tested with PGT?
As genetic medicine has evolved, our understanding of the genetic basis of diseases has increased exponentially.
Over 100 genetic diseases can be tested from a single tube of blood. Many of these are extremely rare; however, common diseases such as cystic fibrosis, Tay-Sachs disease, fragile X syndrome, and others are important to identify in couples seeking to conceive.
BRCA, or the “breast cancer” gene, is another important disease that can be tested for via genetic testing.
How can genetic testing help patients just by knowing that they’re carrying that gene?
If an individual is found to carry a gene that predisposes them to a certain disease, such as cancer, early intervention may help prevent severe forms of the disease.
For couples looking to conceive, many of these conditions are recessive, which means that they do not show symptoms of the disease but merely carry it.
Since we have two copies of each gene, a good copy can protect an individual and prevent disease. When both members of a couple are carriers of a disease, there is a 25% chance they will both contribute the bad copy to their child, which will result in illness.
Where do I go for genetic testing?
Your fertility clinic can perform your genetic testing for you. Most fertility clinics use a reference laboratory that specializes in genetic testing they send your bloodwork to. There are several commercial laboratories available around the U.S.
Preimplantation Genetic Testing for Aneuploidy (PGT-A) is s platform that can accurately determine if an embryo possesses the right number of chromosomes (euploidy) or too few or too many (aneuploidy) with more precision than the vast majority of other platforms.
This is important because published data suggests 50-70% of miscarriages are due to aneuploidy. Selecting the best embryo with PGT-A can substantially reduce these risks.
Preimplantation Genetic Testing for Aneuploidy (PGT-A) is performed by highly specialized laboratories that have expertise in extracting this information from the small amount of DNA that can be obtained from an embryo biopsy.
It is important to select an IVF clinic with extensive experience in this area as it is more complicated than typical genetic testing.
Is genetic testing covered by health insurance?
Most health insurances cover basic genetic testing for couples looking to conceive. Each insurance plan is different, so it is best to check with your insurance provider.
At RMA, your financial counselor will check with your provider for you, so there is one less thing for you to worry about.
What criteria do experts use when referring patients for genetic testing?
At RMA, we offer every couple looking to conceive the opportunity to speak with a genetic counselor who is best qualified to discuss which diseases a couple might be at risk of carrying and what tests they should consider.
What do genetic counselors do?
Genetic counselors are specially trained healthcare professionals with backgrounds in medical genetics and counseling.
These individuals can identify genetic causes of diseases in individuals presenting with a disease. They can also identify families that carry a genetic disease or couples at high risk of having a child with a genetic disease.
Genetic counselors will speak with patients, asking them questions about their family history and ethnic background to identify what they may be at risk of carrying.
What are the advantages and disadvantages of genetic testing?
The main advantage is that early detection may prevent more severe forms of a disease or prevent a couple from having a sick child.
The main disadvantage is that it may cause psychological stress to an individual if they were not previously aware of an increased risk of developing a disease that has no cure.
What if I get bad news back from my genetic test?
Part of ordering a test is knowing what to do with the results.
Early detection of a disease is a good thing. There may be steps that you can take to prevent the disease from progressing or at least help with its symptoms.
Genetic testing on an embryo can prevent the birth of a child with fatal genetic conditions.
If you are struggling with the news you received from your genetic test, we offer emotional support through our counseling support staff.
What is gene therapy?
Gene therapy refers to a series of experimental therapies aimed at using genetic manipulation of the DNA in our cells to treat disease.
In most cases, it would involve inserting new copies of a gene into cells to replace mutated or missing copies of a gene. In other cases, it might involve inserting new genes aimed at targeting a specific disease or turning off a gene that is functioning improperly.
At present, this is highly experimental, very risky, and has not been proven to be effective in any condition.
What are these new DNA sequencing tools?
The technology used in genetic testing has evolved dramatically in the past decade.
PCR (polymerase chain reaction) is a process where specific DNA sequences can be investigated and amplified – this requires knowing the DNA sequence you are looking for. The basis of most genetic testing involves this PCR method.
Sequencing allows us to actually look at the DNA sequence, essentially seeing the entire DNA code.
In the past, it has been a time-consuming and expensive process called Sanger Sequencing.
A new set of technologies called Next Generation DNA Sequencing uses semiconductors to streamline the process by using “chips,” which allow for the simultaneous processing of many samples.
Computers are then used to interpret the massive amounts of data generated to allow for the interpretation of these results. It is a promising new technology that is still developing. Studies are still being performed to see if it is as accurate as traditional techniques like Sanger Sequencing and PCR.
What is whole-genome testing?
Whole-genome testing refers to the idea that the entire DNA sequence (all of our genes) might be scanned to look for genetic mutations.
At present, we are able to sequence a person’s entire genome – meaning we can obtain specific DNA sequences. Unfortunately, interpreting this information is extremely difficult.
Only a small percentage of genes are understood well, and the majority of our DNA is still a mystery to us. This field is expanding rapidly, and new genetic tests and associations are becoming available.





